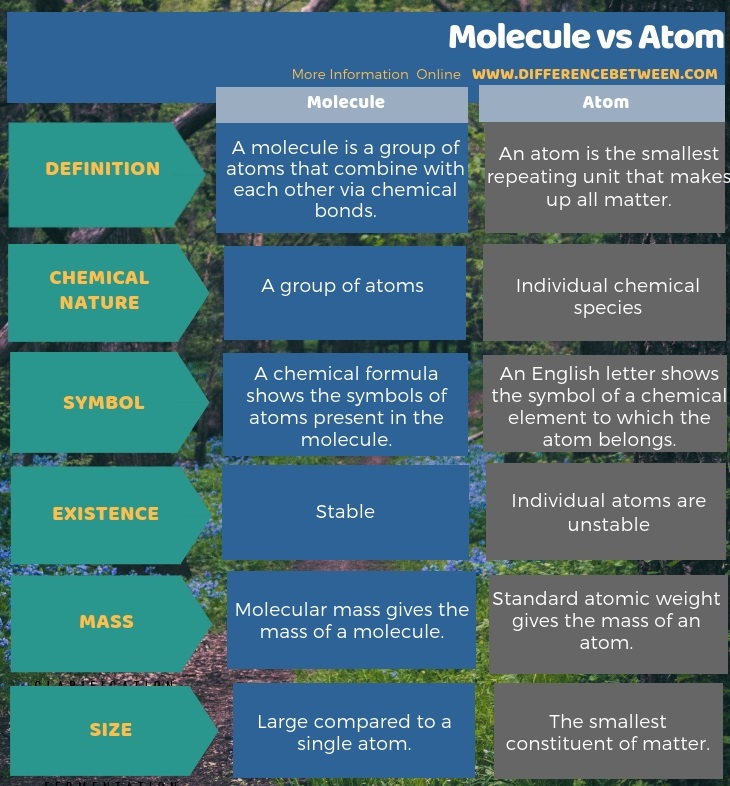 
The electromagnetic force of the protons wants to push them away from each other. Protons are composed of two up quarks and one down quark (uud), and neutrons are composed of one up quark and two down quarks (ddu), as shown in Figure 1.28.There are several forces at work inside the nuclei of atoms. Two of the most common types are the up quark (u) and the down quark (d). Protons and neutrons are composed of smaller particles yet, called quarks. As it turns out, there is a series of more fundamental, or elementary, particles, from which all matter in the Universe is made. While we often think of atoms as the building blocks of matter, they are themselves composed of the subatomic particles protons, neutrons, and electrons. For this reason, the mass numbers shown are not integers. In Figure 1.27, the mass number reported is a weighted average over all the different isotopes for that species. For example, the isotope of helium with just one neutron is abbreviated helium-3 or 3He the more common form of helium, used in helium balloons, is abbreviated helium-4 or 4He. Isotopes of an element are often abbreviated with the element’s name or symbol and the mass number. Both of these isotopes of helium have two electrons. As another example, helium usually has two protons and two neutrons (atomic number 2, mass number 4), but there is an isotope of helium that has two protons and one neutron (atomic number 2, mass number 3). Both of these isotopes of hydrogen have a single electron orbiting the nucleus (whose negative electric charge balances the positive charge of the proton). An isotope of hydrogen that has one proton and one neutron in the nucleus is called deuterium (atomic number 1, mass number 2). For example, the most common form of hydrogen has just a single proton for a nucleus (atomic number 1, mass number 1). Two atoms with the same number of protons but a different number of neutrons are known as isotopes of the same element. In either case, the atom will have a net electrical charge and is then called an ion. In some cases, the atom can lose some of its electrons, or more rarely, gain extra electrons. Since the positive electric charge of the proton has the same strength as the negative electric charge of the electron, the total charge of an atom is zero. The atomic number of an atom determines its chemical properties, because each proton is paired with an electron orbiting the nucleus, and the outer electrons of an atom determine the atom’s chemistry. 
In the interest of space, we have not included atomic numbers 58-71 and 90-103. Letters denote atomic symbol, number above denotes atomic number, and number below denotes atomic mass. Figure 1.27: Periodic table of the elements. All known elements are shown in this table, including many that are familiar to you such as carbon (C), oxygen (O), and iron (Fe), and some that may be less familiar because they occur less frequently in nature, for example, lithium (Li) and beryllium (Be). The mass of an atom depends almost entirely on the number of protons and neutrons the electrons are so much lighter that their contribution to the mass is negligible. It organizes all of the known elements, and this version gives the symbol for each element and its atomic number (number of protons) and atomic mass (number of protons plus neutrons, averaged over different forms of each atom called isotopes - see below). Figure 1.27 shows the Periodic Table of the Elements. Changing this number would mean changing the element and would require a nuclear reaction to do so. The type of atom, or chemical element, depends on the number of protons it contains-this is called its atomic number. 
To put this into perspective, if the nucleus were half the length of an American football field, the size of an atom would be about the distance across the continental United States. An atom is about 10 ⁻10 m in size, while the nucleus, at 10 ⁻15 m, is 100,000 times smaller. Since the electron cloud is much larger than the nucleus, most of an atom is empty space. An atom is composed of a dense central nucleus made up of protons and neutrons, which is surrounded by an electron cloud. Our bodies, the air we breathe, and the planets and stars are all composed of atoms. Lourdes: That’s right. Neutrons are part of atoms too. Keiko: No, electrons and protons make up atoms-atoms are bigger than either electrons or protons. Atoms make up electrons and protons, is that right? Jue: I am confused by all these different particles. Three students are studying for their upcoming exam.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
